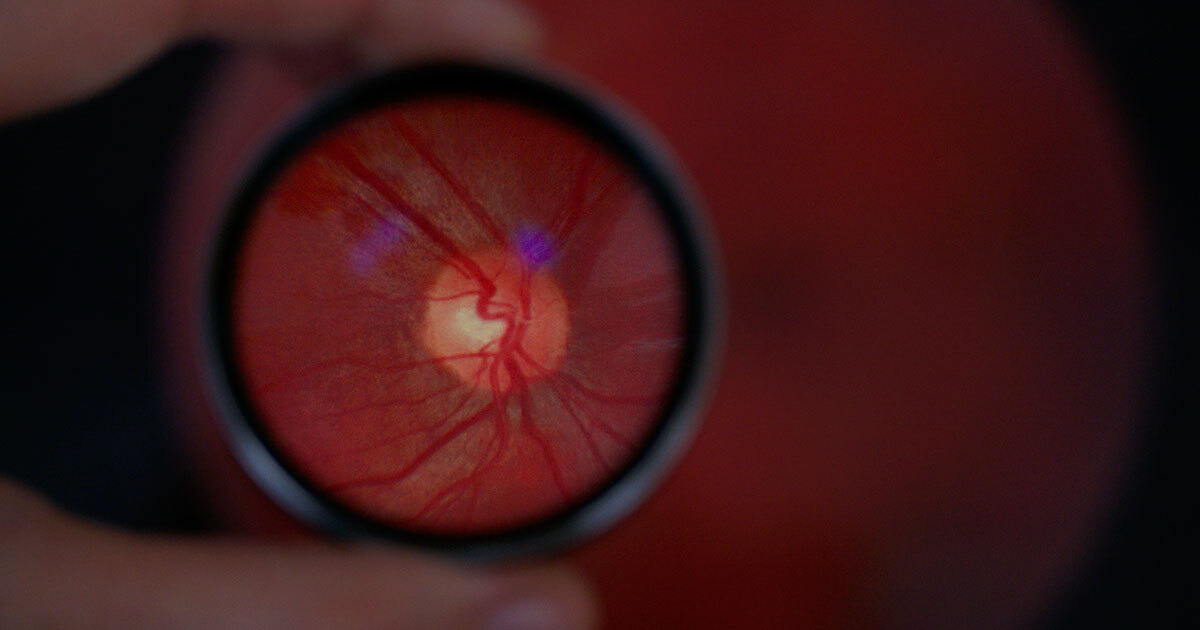
The retina is much more than just an organ of vision: it provides a unique window into the overall health of the body, brain, and heart. Based on this assertion is the work of the ÉTS–DIAGNOS Chair in Artificial Intelligence for Medical Imaging Analysis, led by Professor and Researcher Ismail Ben Ayed.
The result of a partnership between ÉTS and Montreal-based DIAGNOS company, this Chair will push the current boundaries of artificial intelligence to improve early detection and diagnostic support for eye diseases, including numerous systemic diseases. By leveraging the vast array of available retinal images and the power of AI, the objective is to make optical imaging a powerful tool for global health, promoting a more preventive, personalized, and accessible approach to medicine.
An urgent need for quick and reliable testing
Eye diseases are extremely common: over 400 million people worldwide are at risk of developing blindness. Yet, many of these conditions, such as diabetic retinopathy, could be detected much earlier through simple images of the back of the eye. But there are not enough eye care professionals—optometrists and ophthalmologists—and wait times for appointments can allow the disease to progress before a patient can undergo a medical examination and receive a diagnosis.
This is where patient triage becomes essential. The idea is not to replace specialists, but to provide them with tools that quickly identify priority cases using images captured by standard cameras, already in use by optometrists and ophthalmologists.
Seeing Beyond What Meets the Eye
The mission of the ÉTS–DIAGNOS Chair extends far beyond eye diseases. It is based on a fundamental observation: the eye is a unique window into the overall health of the body, brain, and heart. The retina provides direct, non-invasive access to the microcirculation and the nervous system, making it an exceptional source of information on many diseases.
Thanks to the application of artificial intelligence to retinal imaging, we can now detect early signs of not only eye diseases, but also age-related conditions, degenerative neurological disorders, systemic and cardiovascular diseases, and complications related to myopia.
By identifying subtle biomarkers—often invisible to the naked eye during traditional examinations—these technologies pave the way for a more preventive and personalized medical approach that is better integrated into the care pathway. The Chair’s objective is clear: “To make retinal imaging a true tool for global health, for the benefit of patients and healthcare systems,” notes Dr. Ismail Ben Ayed, holder of the Chair.
Algorithms that explain their decisions
Unlike AI approaches considered “black boxes,” the algorithms developed under the Chair do more than merely generate a score or a diagnosis. They are designed to be interpretable: they can indicate which areas of the image led to a decision, such as the presence of specific lesions or vascular abnormalities.
These interpretability mechanisms are essential for clinical adoption. They allow human experts to verify, understand, and trust AI recommendations. The models are also capable of assessing their own uncertainty, flagging cases requiring further validation by a specialist.

Learning with few annotations but lots of data
One major challenge in medical imaging is the scarcity of annotated data: each annotation requires time and clinical expertise. To overcome this, the team is focusing on semi-supervised and weakly supervised learning methods.
In practice, the models are trained using hundreds of thousands—or even close to a million—retinal images, only a small fraction of which are annotated. Unannotated images come from multiple clinics and various devices, making the models more robust against real-world variations.
Foundational models at the heart of the project
The project builds on recent advances in foundational models, which have proven highly effective in computer vision and natural language processing. These models are first trained on large sets of unannotated data to learn general patterns: for example, by masking parts of an image and asking the network to reconstruct them.
Once this phase is complete, the model can be adapted to precise tasks, such as detecting a specific disease using only a few hundred annotated images. This approach significantly reduces costs while improving the system’s ability to adapt quickly to new contexts, devices, or populations.
From research to clinical deployment
Diagnos, an industry partner of the Chair, develops software for optometry and ophthalmology clinics. The company is currently undergoing approval processes with regulatory bodies such as the FDA, Health Canada, and European agencies.
Research conducted at ÉTS plays a key role in ensuring that these algorithms are effective, robust, interpretable, and clinically acceptable—essential criteria for their practical application in medical settings.
Tangible benefits for public health
Ultimately, this research program could revolutionize screening for eye diseases: enabling low-cost population-based screenings, accelerating diagnoses, reducing the risk of blindness, and even preventing some cardiovascular complications. It could also help reduce healthcare costs.
Ismail Ben Ayed is a member of itechsanté, the ÉTS research institute for innovation in health technologies. To learn more about the institute, its mission, themes, flagship projects, and more, visit itechsanté



